|
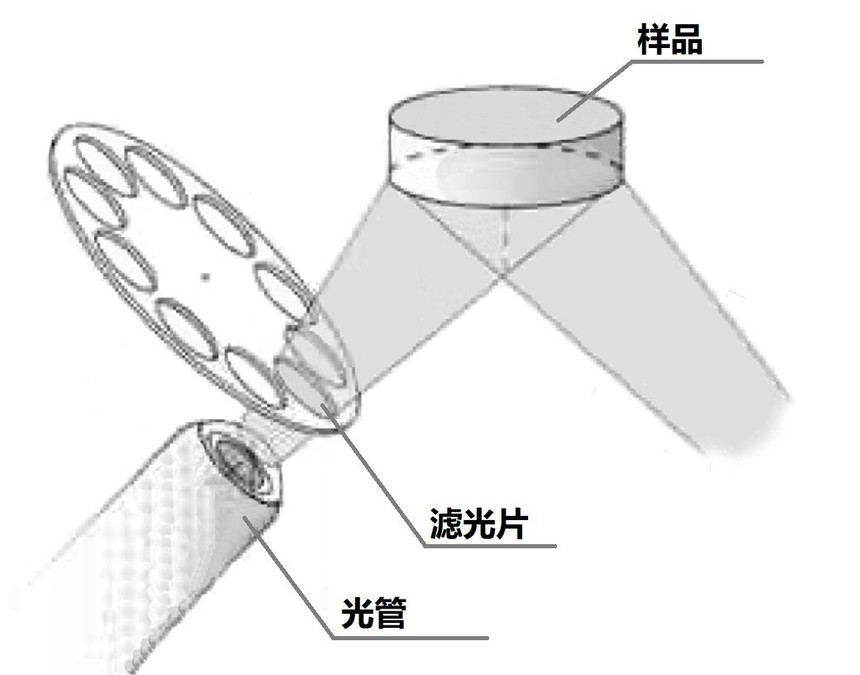
What influence does the filter of a handheld spectrometer have on the sample measurement results?In the parameter table provided by handheld spectrometer manufacturers, there is a parameter called "filter", which often includes the names of some elements, such as aluminum (Al), tungsten (W), etc. During the process of testing materials such as aluminum alloy and soil, one often hears a "sizzling ~ sizzling ~" sound. This is actually the sound of the motor inside the handheld spectrometer rotating, switching between different filters. So, what impact do filters have on the materials and elements we test? The structural schematic diagrams of the light tube, filter and sample in the KMX-RAY handheld spectrometer K-1688
The simple principle of X-ray fluorescence is that the light tube emits X-rays, which are irradiated onto the surface of the sample to produce fluorescence. The detector detects the fluorescence emitted by the sample and determines which elements it contains and their contents based on its characteristics. The X-rays emitted from the X-ray tube play a very important role in the test results. First of all, the X-ray tube determines which elements this handheld X-ray fluorescence spectrometer can detect. Introduction to KMX-RAY XRF Knowledge: Atoms possess wave-like and particle-like properties. The wave-like nature of each element is represented by the wavelength in nm, and the particle-like nature is expressed by the energy in KeV. The handheld spectrometer measures particle elements. The following picture is a periodic table of elements represented by the energy of element particles. The further back an element goes, the higher its energy.
The X-rays emitted by the light tube are not infinite but have a fixed energy. For example, if the light tube voltage of the KMX-RAY K-1688 is set at 35KeV, then in the above figure, if one of the four energies around an element is less than 35KeV, this element can be detected by the K-1688 handheld spectrometer. If all four energies are higher than the voltage set by the light tube, then this element cannot be measured. However, being able to measure does not mean it can be accurate or low. For some samples with very low content, there should be a way to reduce the noise of the detected elements so that the detected elements can be clearly distinguished. For elements with relatively high content but with requirements for detection stability, there must be a way to increase the signal of the element so as to ensure stability. So the filter is a very good means to solve the problem. The following figure shows the X-ray tube spectra obtained by using Ag target tubes with different filters. The spectrogram numbered 1 was obtained without using a filter. Due to the Ag target, there is a very high peak in the low-energy region on the far left. This peak affects the detection limits of elements such as magnesium (Mg), aluminum (Al), and silicon (Si). Only when the peak of an element is higher than the line numbered 1 can it be read by the instrument as having a peak for that element, and such a high peak often indicates a relatively high content. So it is very difficult to measure the low content of light elements with an Ag target light tube without a filter. However, on the contrary, because the energy of this section of the light tube is very strong (compared to numbers 2 and 3), elements such as magnesium (Mg), aluminum (Al), and silicon (Si) will be well excited. Therefore, materials like aluminum alloys that contain high levels of magnesium (Mg), aluminum (Al), and silicon (Si) are very suitable for detection without filters.
It is also necessary to look at the graphs numbered 2 and 3. Graph numbered 2 shows the light tube spectra measured with a relatively thin Ag as the filter, and graph numbered 3 shows the light tube spectra measured with a relatively thick Ag as the filter. We can clearly see that in the energy range of 10-13 KeV, the spectra numbered 2 and 3 are significantly lower than those numbered 1. Then, for the detection of low Pb content (with energies of 10.55 and 12.61 KeV), choosing Ag filters is quite appropriate. If it is an application such as detecting whether heavy metals in the soil exceed the standard, choosing a suitable filter is very important. For the detection of different samples, it is actually necessary to adopt different filters based on the elements that users are concerned about. For instance, when testing stainless steel, should the focus be on whether the nickel (Ni) element meets the standards or on the titanium (Ti) element? This requires handheld spectrometer manufacturers to understand the users' demands before delivering the instruments and accordingly design different filter and tube voltage combination schemes. For more information about the spectra obtained by testing different samples with different filters, you can search "KMX-RAY" across the entire web. |